Invention Summary:
The drive to replace precious platinum group metal (PGM)-based oxygen reduction reaction (ORR) catalysts has boosted the research and development of PGM free single-atom catalyst. However, numerous challenges remain in large scale practical application, efficiency, and long-term durability.
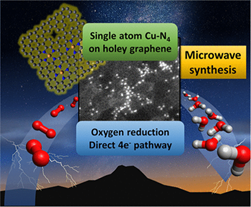
Rutgers Researchers have developed a time and energy efficient microwave assisted approach for the direct fabrication of electrochemical single-atom catalysts (E-SAC) without the requirement of post-treatment. The as-prepared Cu-E-SAC, where Cu-active centers were incorporated in the holey graphene nanoplatelets, exhibited excellent catalytic activity and selectivity in reducing oxygen to water in both acidic and alkaline media. The selectivity toward the desired direct 4e- pathway to water in acidic media is even higher than that in alkaline media, thus suitable to be incorporated with the current proton-exchange membrane (PEM) technologies.
Advantages:
- Time and energy efficiency
- No post-treatment is required
- Densely and uniformly dispersed active catalytic sites
- Low metal atom segregation
Market Applications:
- Proton-exchange membrane fuel cells
- Catalysts
Intellectual Property & Development Status: Patent pending. Available for licensing and/or research collaboration.
Publication: Li et al. ACS Appl. Energy Mater. 2020, 3, 9, 8266–8275