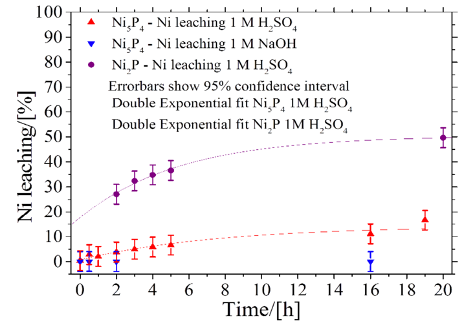
Amount of Ni in the electrolyte during chronopotentiometric (CP) analysis in both acid and alkali. For comparison Ni2P in acid is also shown
Invention Summary:
Hydrogen is a versatile energy carrier and a valuable bulk chemical used for storing intermittent renewable energy and it also serves as a major chemical feedstock. Hydrogen can be produced from a variety of resources, electrochemical water splitting being one of them. This technique requires the use of electrolyzers containing Platinum (Pt) or Nickle (Ni) electrodes. Pt electrodes provide high efficiency; however, they are very expensive. On the other hand, Ni electrodes have low efficiency but are widely used owing to its low cost.
In order to address this issue Rutgers researchers have developed a novel electrocatalyst containing nickel organophosphine molecular complexes (used here, Ni5P4) for hydrogen generation by water splitting. Electrodes coated with Ni5P4 electrocatalyst exhibit low onset overpotential in both acid and base and outperforms other non-noble catalysts. Additionally, this electrocatalyst demonstrate a stable hydrogen evolution reaction (HER) activity in both acidic and alkaline pH and provides the benefit of cost-effective renewable H2 production from water when paired with water oxidation catalysts.
Advantages:
- Exceptional chemical stability in strong acid and strong base.
- Stable hydrogen evolution reaction activity in acidic and alkaline medium.
- Cost-effective renewable H2 production.
Market Applications:
- Hydrogen production
- Intermittent renewable energy storage
Intellectual Property & Development Status: