
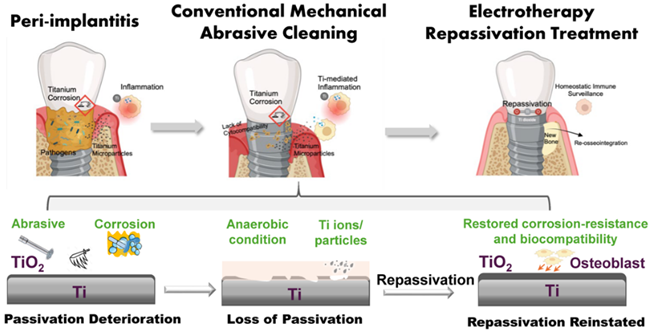
In Situ electrochemical repassivation therapy of titanium dental implants can restore corrosion resistance and enhance cellular biocompatibility.
Invention Summary:
Peri-implantitis affects up to 25% of dental implant patients and is a leading cause of implant failure, representing a global market burden exceeding $1B annually. Current treatments focus on decontamination but fail to address the underlying material failure of titanium implants: progressive corrosion driven by inflammation. The present gold standard antimicrobial treatments fail to treat >50% of cases highlighting a substantial unmet clinical need and positioning the field as a strong candidate for FDA Breakthrough Device designation.
Rutgers researchers have developed a first-in-class, in situ electrochemical medical device that restores titanium surface integrity. By enabling in situ repair of the implant’s passivation layer, the device significantly improved anti-corrosion performance, effectively hindering titanium degradation to alleviate the corrosion-inflammation cycle of titanium dental implants.
Market Applications:
- Treatment and management of peri-implantitis in dental patients
- Maintenance and longevity enhancement of titanium dental implants
- Advanced implant care from dental clinics and oral healthcare providers
Advantages:
- Only therapy that directly restores the titanium oxide passivation layer in vivo
- Interrupts the corrosion–inflammation cycle rather than treating symptoms
- Chairside procedure compatible with all existing titanium implant systems
- The technology is positioned for translational development toward chairside clinical use, with a clear regulatory and commercialization pathway in implant dentistry
Proof of Concept & Validation:
- Demonstrated restoration of titanium corrosion resistance under inflammatory conditions
- Proved cellular biocompatibility following in situ electrochemical repassivation according to ISO standards
- Supported by preclinical data from a leading academic implantology group
Intellectual Property & Development Status: PCT application filed. Patent pending. Available for licensing and/or research collaboration. For any business development and other collaborative partnerships, contact: marketingbd@research.rutgers.edu